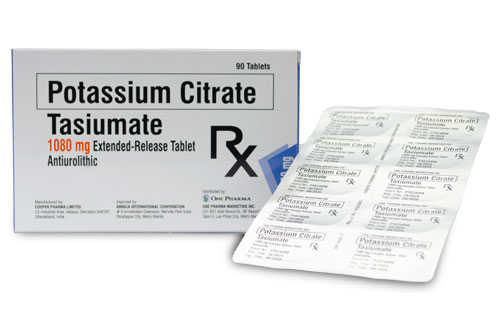
Potassium Citrate
TASIUMATE
Regulatory Class: Rx
Generic Name: Potassium Citrate
Formulation/s: 1080 mg of Potassium Citrate (10 mEq), USP
Indications or Use: : Management of renal tubular acidosis (RTA) w/ Calcium stones, hypocitraturic Calcium oxalate nephrolithiasis of any etiology & uric acid lithiasis w/ or w/o Calcium stones.
Dosage and Directions for Use: Severe hypocitraturia (urinary citrate <150 mg/day) Initially 60 mEq/day (20 mEq thrice a day or 15 mEq 4x a day). Mild to moderate hypocitraturia (>150 mg/day) Initially 30 mEq/day (10 mEq thrice a day).
Administration: Take with or without food. Swallow whole, do not crush/suck/chew.
Contraindication: Hyperkalemia (or who have conditions predisposing to hyperkalemia). Patients suffering from delayed gastric emptying, esophageal compression, intestinal obstruction or stricture or those taking anticholinergic medication. PUD. Renal insufficiency (GFR <0.7 mL/kg/min).
Special Precaution: : Avoid in patients w/ chronic renal failure or any other condition which impairs Potassium excretion e.g., severe myocardial damage or heart failure. Avoid concomitant use w/ Potassium-sparing diuretics (e.g., triamterene, spironolactone or amiloride). Pregnancy & lactation. Children.
Adverse Reaction: Abdominal discomfort, vomiting, diarrhea, loose bowel movements or nausea.
Interactions: Severe hyperkalemia w/ K-sparing diuretic (e.g., triamterene, spironolactone or amiloride).
Presentation/ Packaging: : Extended-Release tablet 1,080 mg x 90’s.
For more information, please see full product information.