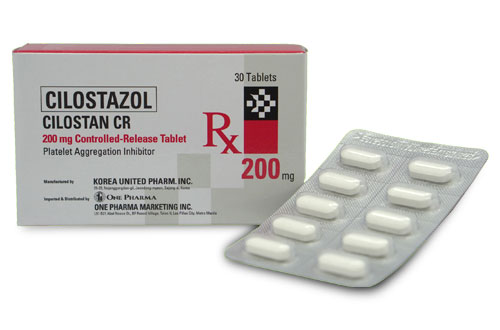
Cilostazol
CILOSTAN CR
Regulatory Class: Rx
Generic Name: Cilostazol
Formulation/s: 200 mg Controlled Release Tablet
Indications or Use: Treatment of ischemic symptoms including ulcer, pain & coldness of the extremities in chronic arterial occlusion. Prevention of reoccurence of cerebral infarction (excluding cardiogenic cerebral embolism).
Dosage and Directions for Use: 200 mg once daily.
Administration: Take without food. Take at least 30 min before or 2 hours after meals.
Contraindication: Hypersensitivity. Known predisposition to bleeding (e.g., active peptic ulceration, haemorrhagic stroke, proliferative diabetic retinopathy, poorly controlled hypertension). Coronary Heart Failure, women of childbearing potential and pregnancy.
Special Precaution: Patients using anticoagulants (e.g., warfarin), platelet aggregation inhibitor including aspirin, ticlopidine & thrombolytics including urokinase & alteplase; prostaglandin E1 or derivative (alprostadile, rimaprodex, alphadex). Patients w/ menstruation. Severe renal impairment (CrCl ≤25 mL/min). Moderate or severe hepatic impairment. Known predisposition to bleeding (e.g., active peptic ulceration, haemorrhagic stroke, proliferative diabetic retinopathy, poorly controlled hypertension). History of ventricular tachycardia, ventricular fibrillation or multifocal ventricular ectopics; prolongation of QTC interval. Report any episode of bleeding or easy bruising; early development of dyscrasia. May cause dizziness affecting ability to drive or operate machinery. Pregnancy & lactation. Elderly.
Adverse Reaction: Encephalorrhagia, pneumorrhagia, fundus bleeding, nasal hemorrhage, hematuria. Pancytopenia, agranulocytosis. Interstitial pneumonia accompanied with flush, cough, dyspnea, disorder of chest, eosinophilia. Coronary Heart Failure, cardiac infarction, angina pectoris, ventricular tachycardia.
Interactions: May cause bleeding with anticoagulants e.g., warfarin; platelet aggregation inhibitors e.g., aspirin, ticlopidine & thrombolytics including urokinase & alteplase; prostaglandin E1 or derivatives (alprostadile, rimaprodex, alphadex). May increase blood levels w/ CYP3A4 inhibitors (erythromycin, cimetidine, grapefruit juice; omeprazole); organic of CYP3A4 (diltiazem).
Presentation/ Packaging: Controlled Release 200 mg x 30’s
For more information, please see full product information.