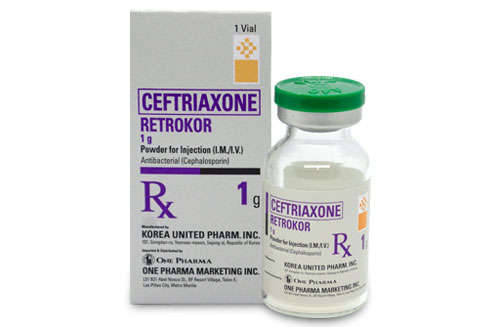
Ceftriaxone
RETROKOR
Regulatory Class: Rx
Generic Name: Ceftriaxone Sodium
Formulation/s:
Indications or Use: Respiratory infections e.g., pneumonia, bronchitis, otorhinolaryngology infections, renal or urinary tract infections, meningitis, bone & joint, skin, wound & soft tissue, GIT, genital infections including gonorrhea, peritonitis, cholecystitis, cholangitis, peri-op prophylaxis of infections & in patients w/ impaired immunologic function.
Dosage and Directions for Use: Adult & children >12 yr 1-2 g once daily IM/IV, up to 4 g daily. Neonate & children (15 days-12 yr) 20-80 mg/kg once daily. Neonate (w/in 14 days) 25-50 mg/kg once daily. Meningitis Neonate & children 100 mg/kg once daily. Max: 4 g/day. Duration of treatment for N. meningitidis 4 days, H. influenzae 6 days, S. pneumococcus 7 days. Gonorrhea 250 mg IM. Peri-op prophylaxis 1-2 g given 30-90 minute before surgery.
Contraindication: Hypersensitivity to cephalosporins, penicillin, anilide local anesthesia (e.g., lidocaine). History of shock.
Special Precaution: Patients w/ history of allergy to drugs. Patients whose family is susceptible to cause the allergy symptoms e.g., bronchial asthma, eruption & urticaria. Severe renal disorder. Patients who cannot ingest orally or parenteral patients, elderly & patients w/ systemic bad condition. Perform regular liver, renal, hematologic tests.
Adverse Reaction: Shock, hypersensitivity reactions, hematologic, hepatic & renal effects. GI effects e.g., severe colitis. Resp disorders, superinfection, avitaminosis.
Interactions: Furosemide, aminoglycosides, alcohol, chloramphenicol.
Presentation/ Packaging: Powder for injection (vial) 1 g x 1’s
For more information, please see full product information.