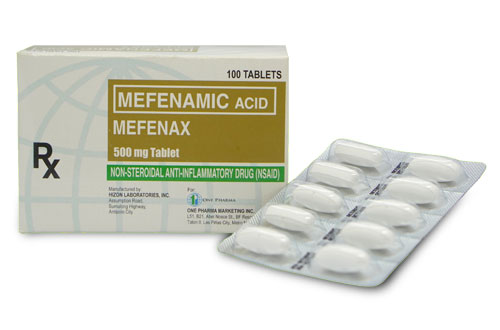
Mefenamic Acid
MEFENAX
Regulatory Class: Rx
Generic Name: Mefenamic Acid
Formulation/s: 500 mg tablet of Mefenamic Acid, BP
Indications or Use: For the relief of pain including muscular, arthritic conditions, traumatic, dental post-surgical and postpartum pain; headache, and fever. Also, for the relief of primary dysmenorrhea.
Dosage and Directions for Use: Adult dose: 500 mg every 8 hours or as prescribed by a physician. Treatment should not be continued for more than 7 days.
Contraindication: Patients with ulceration or inflammation of the gastrointestinal tract.
Special Precaution: Pregnancy and Lactation. Since there are no adequate and well-controlled studies in pregnant women, this drug should be used only if the potential benefits to the mother justify the possible risk to the fetus. It is not known if mefenamic acid or its metabolites cross the placenta. However, because of the effects of drugs in this class (i.e., inhibitors of prostaglandin synthesis) on the fetal cardiovascular system (e.g., premature closure of the ductus arteriosus), the use of mefenamic acid in pregnant women is not recommended. Mefenamic acid inhibits prostaglandin synthesis which may result in prolongation of pregnancy and interference with labor when administered late in the pregnancy. Women on mefenamic acid therapy should consult their physician if they decide to become pregnant. Inhibition of prostaglandin synthesis might adversely affect pregnancy. Data from epidemiological studies suggest an increased risk of spontaneous abortion after use of prostaglandin synthesis inhibitors has been shown to result in increased pre- and post-implantation loss.
Adverse Reaction: Side effects are few and mild at the recommended dosage for analgesic use and readily detected and easily controlled when encountered with high dosage and long-term use. Gastric irritation is infrequent and may be minimized by taking medications with meals. Diarrhea has been observed when Mefenamic acid was given in higher than recommended doses and/or long-term continuous therapy. Skin rash has occurred but disappeared on withdrawal of medication. Rarely, thrombocytopenia and reversible hemolytic anemia have been reported with Mefenamic acid.
Interactions: Acetylsalicylic acid: Mefenamic acid interferes with the anti-platelet effect of low-dose aspirin, and thus may interfere with aspirin’s treatment of cardiovascular disease. Anticoagulants: Mefenamic acid has been shown to displace warfarin from protein binding sites, and may enhance the response to oral anticoagulants. Therefore, concurrent administration of mefenamic acid with oral anticoagulant drug requires frequent prothrombin time monitoring.
Anti-hypertensives including diuretics, angiotensin-converting enzyme (ACE) inhibitors, angiotensin II antagonists (AIIA) and beta-blockers: NSAIDs can reduce the efficacy of diuretics and other antihypertensive drugs, including ACE inhibitors, AIIA and beta-blockers. In patients with impaired renal function (e.g. dehydrated patients or elderly patients with compromised renal function), the co-administration of an AIIA and/or diuretics with a cyclo-oxygenase inhibitor can increase the deterioration of the renal function, including the possibility of acute renal failure, which is usually reversible. The occurrence of these interactions should be considered in patients taking Mefenamic acid with an ACE inhibitor or an AIIA and/or diuretics. Therefore, the concomitant administration of these drugs should be done with caution, especially in elderly patients. Patients should be adequately hydrated and the need to monitor the renal function should be assessed in the beginning of the concomitant treatment and periodically thereafter. Corticosteroids: Increased risk of gastrointestinal ulceration or bleeding. Cyclosporine: Because of their effect on renal prostaglandins, NSAIDs such as mefenamic acid may increase the risk of nephrotoxicity with cyclosporine. Hypoglycemic agents: There have been reports of changes in the effects of oral hypoglycemic agents in the presence of NSAIDs. Therefore, Mefenamic acid should be administered with caution in patients receiving insulin or oral hypoglycemic agents. Lithium: Mefenamic acid has produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. Thus, when Mefenamic acid and lithium are administered concurrently, patients should be observed carefully for signs of lithium toxicity. Methotrexate: Caution is advised when methotrexate is administered concurrently with NSAIDs, including Mefenamic acid because NSAID administration may result in increased plasma levels of methotrexate, especially in patients receiving high doses of methotrexate. Tacrolimus: Possible increased risk of nephrotoxicity when NSAIDs are given with Tacrolimus.
Presentation/ Packaging: Alu-PVC Blister pack x 10’s (Box of 100’s)
For more information, please see full product information.